
Sarah Grünert, MD
Consultant, Metablic pediatrician
MetabERN subnetwork coordinator
E: sarah.gruenert@uniklinik-freiburg.de
T: +49 (0)761 270-43720
F: +49 (0)761 270-45270
Medical Center – University of Freiburg
Center for Pediatrics and Adolescent Medicine
Mathildenstr. 1
79106 Freiburg
Germany
Our research has two main foci: Fatty acid oxidation disorders and glycogen storage diseases.
The group’s focus is on translational and clinical research in fatty acid oxidation defects. Our main research interests are the long-term outcome of the different metabolic defects, the effects of environmental factors and dietary interventions and the impact of FAODs on the life of affected patients and their families.
RESEARCH THEMES
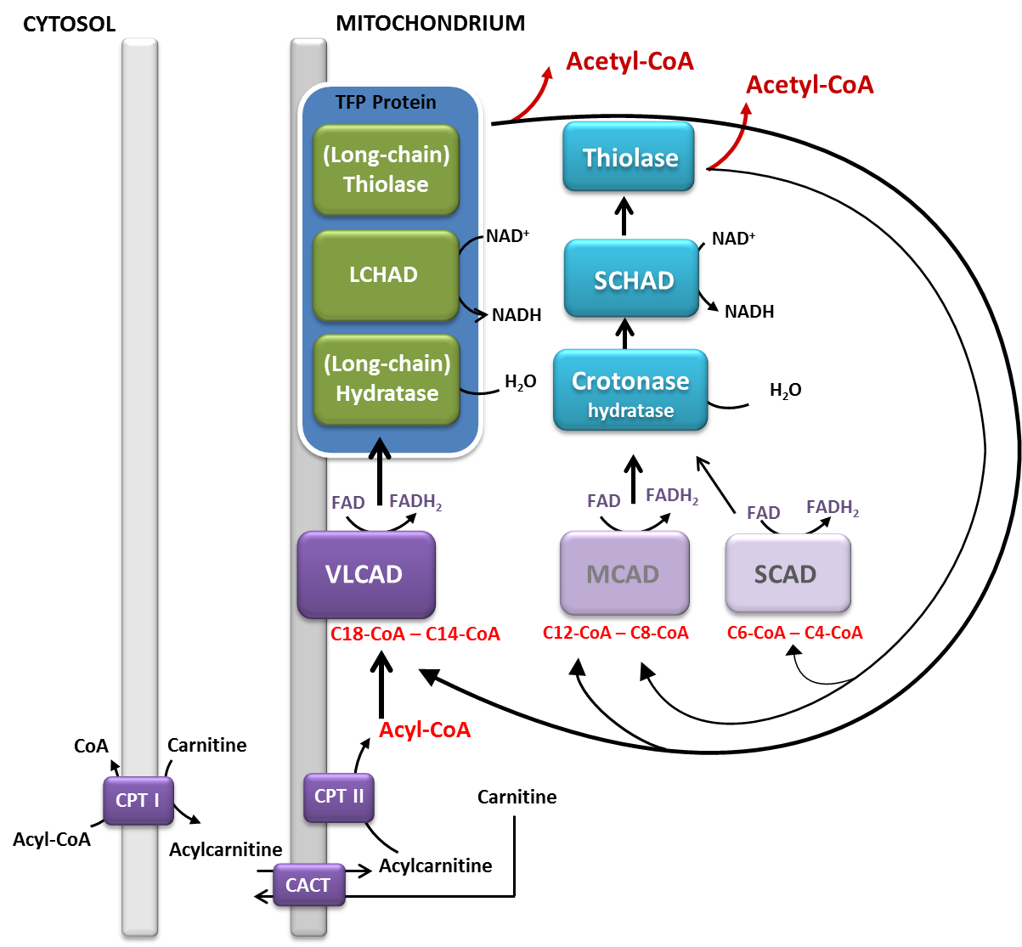
Fatty acid oxidation disorders (FAODs) are a group of inborn errors of metabolism that affect either the transport of fatty acids or their mitochondrial β-oxidation. As a consequence, fatty acids cannot be used as an appropriate energetic source during special conditions, such as prolonged fasting, exercise or other catabolic states. Patients usually present with skeletal myopathy, episodic rhabdomyolysis, hepatopathy and cardiomyopathy, however, the clinical picture is heterogeneous. Many patients receive a fat-reduced and fat-modified diet. FAODs are target diseases of newborn screening programs in many countries, including Germany.
Our work aims at a better understanding of the natural history and long-term complications of these genetic disorders, the effects of dietary interventions and the impact of FAODs on the life of affected patients and their families. We work in close collaboration with the German patient organisation for fatty acid oxidation disorders (Fett-S.O.S e.V.).
Our working group also coordinates the global guideline for the diagnosis and management of long-chain fatty acid oxidation disorders.
TEAM
- Stephanie Nauck (MD student)
- Maren Thiel (MD student)
CURRENT PROJECTS
Long-term outcome of patients with LCHAD/MTP deficiency
Implementation of tandem mass spectrometry-based newborn screening has led to a significant reduction of mortality and morbidity in patients with FAODs. Nevertheless, many patients with LCHAD and MTP deficiency develop long-term complications such as peripheral neuropathy and retinopathy. Current treatment strategies seem to be unable to fully prevent these complications, and the pathophysiology of neuropathy and retinopathy is still not fully understood.
In a multicentre study in cooperation with the metabolic centre Heidelberg we currently assess the long-term outcome of German patients with LCHAD and MTP deficiency.
Life with a child affected by a fatty acid oxidation defect: Impact on different areas of life, psychological aspects and coping strategies
In this study which is carried out in collaboration with the German patient organisation for fatty acid oxidation disorders (Fett-S.O.S e.V.) psychosocial issues of having a child with a long-chain FAOD and the impact of the chronic disease on different aspects of the daily life of parents are assessed.
Effects of medium-chain triglycerides (cooperation projects with PD Dr. Sara Tucci)
Medium-chain triglycerides (MCT) are commonly used in the dietary treatment of long-chain FAODs, however, long-term effects of MCT have scarcely been investigated. Two clinical studies focusing on MCT are currently ongoing. In one project performed in cooperation with the Medical Physics Department Freiburg we evaluate the effects of MCT on the body fat distribution and composition applying magnetic resonance imaging and spectroscopy.
A second study investigates the effects of MCT supplementation on the composition of fatty acids in plasma and erythrocyte membranes of patients with FAODs and healthy controls.
Glycogen storage diseases are a group of rare inborn errors of carbohydrate metabolism. Our research focuses on the elucidation of pathophysiological mechanisms in cell models as well as the impact of hepatic glycogen storage disorders on the life of affected patients.
RESEARCH THEMES
Glycogen storage diseases (GSDs) are a group of inborn errors of metabolism that result from defects in any of the enzymes involved either in glycogen synthesis or glycogen degradation. GSDs can be divided in two major entities: GSDs with hepatic involvement that usually present with hypoglycemia and often hepatomegaly and muscle GSDs with predominantly neuromuscular symptoms such as muscle weakness and/or hypotonia.
Our research focuses on hepatic storage diseases including type I, and the so-called ketotic GSDs, type III, VI, IX, XI and 0, and our projects comprise basic research projects as well as clinical and translational studies. We apply metabolomic methods to investigate effects of defective glycogen degradation on metabolic networks. Clinical studies aim at a better understanding of the long-term complications of GSDs and the impact of these chronic diseases on the everyday life of affected patients.
We work in close cooperation with the German patient organisation for glycogen storage diseases (SHG Glykogenose Deutschland e.V.).
TEAM
- Dr. Luciana Hannibal (PhD, collaborating research group leader)
- Johannes Schmitt (MD student)
- Julia Hinkel (MD student)
- Jule Theimer (MD student)

CURRENT PROJECTS
Mitochondrial dysfunction in ketotic glycogen storage diseases
It has recently been shown in cell and animal models that GSD I is associated with mitochondrial dysfunction. In this project we investigate the energy metabolism of fibroblasts of GSD Ia, Ib, III and healthy controls studying
- mitochondrial organization in live cells by spinning-disc confocal microscopy (mitochondrial content, network morphology)
- oxygen consumption rates
- extra- and intracellular metabolites by targeted LC-MS/MS (metabolomics)
Evaluation of vascular dysfunction in ketotic glycogen storage diseases
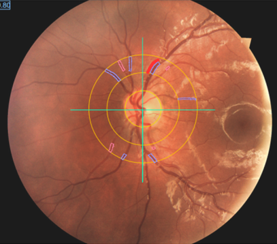
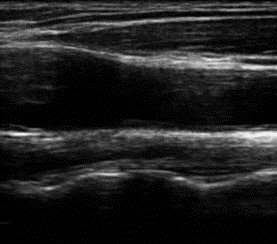
Patients with hepatic GSDs usually have elevated levels of triglycerides and cholesterol in blood. Only few studies have addressed the question if the abnormal lipid concentrations are associated with an elevated risk of vascular dysfunction and premature atherosclerosis. These studies yielded in controversial results. Pathophysiologic factors that may contribute to vascular dysfunction and development of atherosclerosis are oxidative stress and inflammation. In a current clinical study we evaluate the risk of vascular dysfunction in patients with hepatic glycogen storage disorders using different noninvasive techniques (including ultrasound of the carotid artery for the assessment of the functional intima media thickness, investigation of the ocular fundus/retina for the analysis of retinal microvascular diameters and 24h blood pressure measurements) as well as new markers in blood (aminothiols).
Vitamin B12 metabolism in hepatic glycogen storage disease
Patients with GSDs have to stick to a special diet which poses them at risk of vitamin B12 deficiency. On the other hand, liver damage has been described as cause of elevated vitamin B12 concentrations in blood.
This study is the first comprehensive assessment of the vitamin B12 metabolism in hepatic GSD patients.
The aims of this study are
- to assess the vitamin B12 status of patients with hepatic GSDs using different plasma biomarkers of vitamin B12 status, thereby excluding functional deficiency
- to elucidate whether associations exist between vitamin B12 status and liver function/metabolic control, and
- to elucidate which other factors (supplementation, age etc.) influence vitamin B12 levels in this patient cohort.
Glycogen storage disease type I in adulthood
GSD Ia is clinically characterised by severe fasting hypoglycemia, hepatomegaly, failure to thrive, growth retardation, truncal obesity, doll-like facies, short stature, bleeding tendency and hypotrophic muscles. Laboratory findings include hyperuricemia, hyperlipidemia, and elevated lactate levels. GSD Ib shares these main features with GSD Ia, but is additionally aggravated by neutropenia and neutrophil dysfunction, associated with frequent infections and possible chronic inflammatory bowel disease. As patients with GSDs I have to adhere to a strict diet (frequent meals, supplementation of uncooked cornstarch or continuous overnight tube feeds) and have to live with the risk of severe hypoglycaemia, these diseases have a high impact on daily life of patients. Due to improved therapy most patients with GSD I nowadays reach adulthood. As a consequence, they have to deal with new issues of their disease connected with adult daily life such as integration in work and social life, sports, travelling, dietary management, disease monitoring, self-autonomy, family planning, and the patients’ attitude towards their disease.
In this project we investigate the impact of GSD I on adult life in patients from different metabolic centres throughout Germany.
COLLABORATIONS, COOPERATIONS AND NETWORKS
- Glycogen storage disease program, Conneticut Children’s Hospital
- Terry Derks (Department of metabolic Diseases, University Children’s Hospital Groningen)
- Gepke Visser (Department of Metabolic Diseases, Wilhelmina Children’s Hospital, UMCU in The Netherlands (CIMD-UMCU)
- MetabERN (European Reference Network for Hereditary Metabolic Disorders)
- Selbsthilfegruppe Glykogenosen Deutschland e.V. (Self-help group for people with Glycogenosis, scientific advisory board)
- Selbsthilfegruppe für angeborene Fettsäurenoxidationsstörungen Fett-SOS e.V. (Self-help group for people with congenital fatty acid oxidation disorders)
SELECTED RECENT PUBLICATIONS
For a complete publication list for Sarah Grünert, please use the following links:
PubMed: https://www.ncbi.nlm.nih.gov/pubmed/?term=Gr%C3%BCnert+SC