
Professor Brigitte Stiller
Professor, Medical Director
E: brigitte.stiller@universitaets-herzzentrum.de
Department of Congenital Heart Defects and Pediatric Cardiology
University Heart Center Freiburg – Bad Krozingen
Mathildenstr. 1
79106 Freiburg
Germany
These projects focus on developing innovative methods to improve safety and reduce the burden on children with complex congenital heart defects.
CURRENT PROJECTS
Medical devices
Over the past decades, the morbidity and mortality of children with congenital heart defects requiring surgery or intervention has more than halved. For some previously untreatable diagnoses, completely new treatment options have been developed.
We initiated a prospective interventional multi-center clinical trial involving high-risk neonates and infants presenting native or rather re-stenosis after surgical repair and more complex forms of aortic arch obstruction, respectively and were able to confirm the efficacy and safety of the new breakable Osypka Babystent. Unfortunately until now no stent has been certified in the US or Europe for CoA in neonates and infants, so the Osypka Babystent might become the first since this stent offers the chance to overcome the dilemma between the two mandatory requirements: the stent must be on the one hand expandable up to adult size and on the other hand small enough to fit through the femoral artery of a baby. (Funding: industry support from Osypka, Rheinfelden, Germany.)
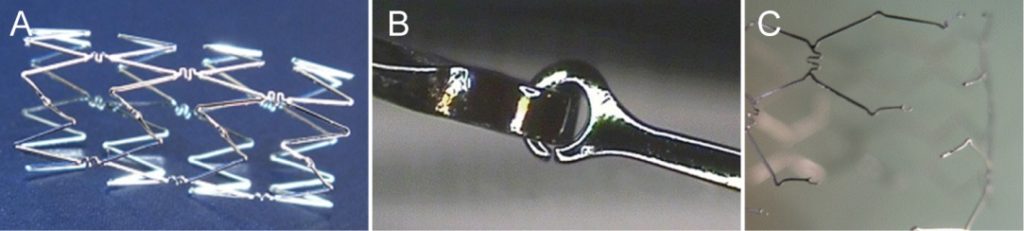
In our clinic, we collaborate in research the further development of various products both on patients and in the laboratory. These include the further development of devices for ASD closure (GSO, Ceraflex). (Funding: industry support from Gore, Lifetech/ Medtonik)
Catheter strategy to ease the procedure and reduce radiation exposure when requiring neck access
We have been able to prove that upside-down positioning of the patient on the catheter table facilitates handling in a straightforward manner when access from the neck is required. It significantly reduces local radiation exposure for the primary operator in the pediatric and, most impressively, in the adult phantom.
Cooperation: Department of Occupational Safety, Medical Center – University of Freiburg.
SELECTED RECENT PUBLICATIONS
Coming soon!